 
National Institute of Standards and Technology. Journal of Quantitative Spectroscopy and Radiative Transfer. "Temperature-dependent mid-IR absorption spectra of gaseous hydrocarbons". Chemical Engineering Research Information Center. ^ "Pure Component Properties" (Queriable database).The standard molar entropy at pressure is usually given the symbol S. These are often (but not necessarily) chosen to be the standard temperature and pressure. ^ Lange's Handbook of Chemistry, 10th ed. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest.Archived from the original (PDF) on 9 October 2006. National Institute of Standards and Technology (15th Symposium on Thermophysical Properties). Rank each set of substances in order of increasing standard molar entropy (S). "Viscosity and Surface Tension of Saturated n-Pentane" (PDF). ^ Lange's Handbook of Chemistry, 10th ed.Table data obtained from CRC Handbook of Chemistry and Physics 47th ed. Structure and properties Structure and properties It is highly recommend that you seek the Material Safety Datasheet ( MSDS) for this chemical from a reliable source such as eChemPortal, and follow its directions. The handling of this chemical may incur notable safety precautions. What is E°(cell) for the reaction 2 Au(s) + 3 Ca²+(aq) → 2 Au³+(aq) + 3 Ca(s)? E° (V) Standard Reduction Potentials 1.5 Au³+ (aq) + 3e → Au(s) Ca²+ (aq) + 2e → -2.This page provides supplementary chemical data on n-pentane. What is K for a reaction if AG° =-77.9 kJ/mol at 25.0 ☌? (R = 8.314 J/mol A) AS is (-) B) AS is (+) C) AS = 0 D) AS cannot be predicted for this reaction.ĭetermine AG for 2 NO₂(g) → N₂O4(g) at 25☌. A table like this can be used in much the same way as a table of standard enthalpies of formation in order to find the entropy change Sm for a reaction occurring at standard pressure and at 298 K. Cl₂ → 2Cl(g) Predict the change in entropy (AS) of this reaction. Values of the standard molar entropies of various substances at 298 K (25C) are given in the table. It can be decomposed to form two Cl atoms as shown below. E) Negative, because there are more moles of reactant than product.Ĭl₂ is a stable diatomic molecule. D) Negative, because there are more moles of gas on the reactant side than the product side. Definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for standard enthalpy and Gibbs free energy of formation, as well as standard entropy and molar heat capacity, of 370 inorganic compounds. 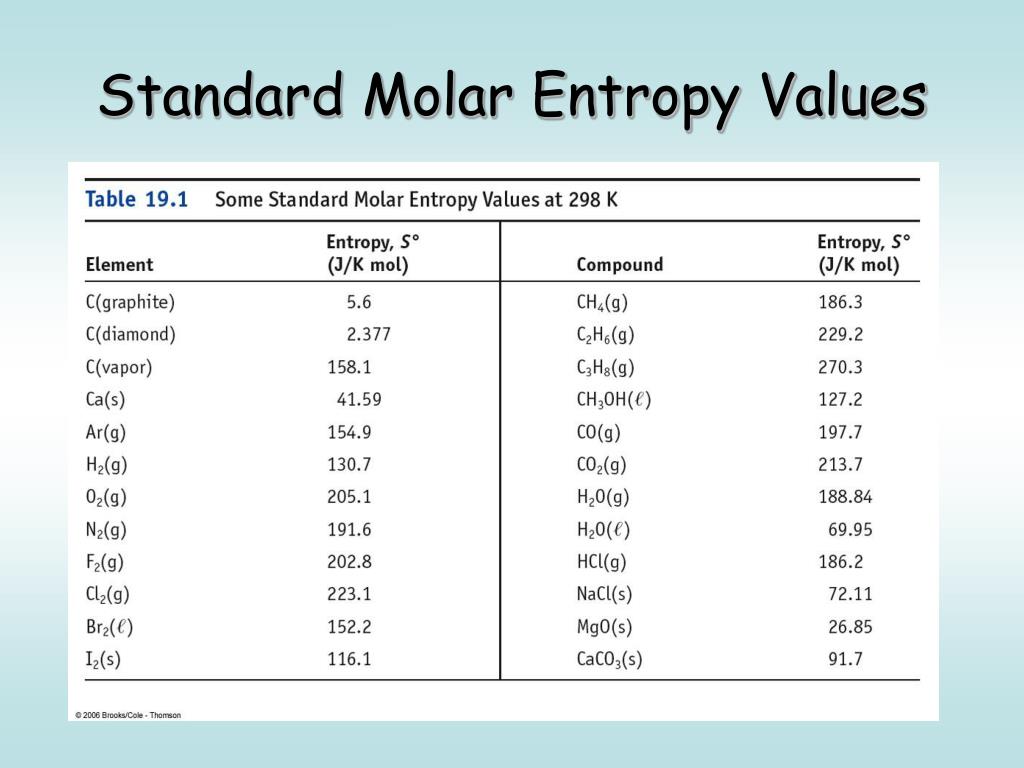
C) Positive, because it is a synthesis reaction. B) Positive, because there are more moles of reactant than product. What will the sign on AS be for the following reaction and why? 2 Mg (s) + O₂(g) → 2 MgO (s) A) Positive, because there is a solid as a product. Standard molar entropy The total (minimal) amount of entropy that 1 mole of a substance gains, as it is brought from 0K to the standard conditions is known. Which of the following has the largest standard molar entropy, Sº (298.15 K)? A) He(g) B) H₂(g) C) NaCl(aq) D) KBr(s) E) Hg(1) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
